The Complete Guide for Food Business Owners & Manufacturers in India
“Selling a food product without expert food ingredient assessment and food formulation review is like launching a ship without checking for leaks – costly, risky, and entirely avoidable.”
If you are a food business owner or food manufacturer in India, you already know that launching a food or nutraceutical product is not just about having a great recipe. It is about ensuring that every ingredient in your product is legally permitted, safe, compliant with FSSAI regulations, and correctly proportioned as per the regulation. This is precisely where food ingredient assessment and food formulation review become your most important regulatory tools before launching or spend a single rupee on production.
India’s food industry is one of the fastest-growing sectors in the world, projected to reach USD 1.2 trillion by 2027. Yet, thousands of food businesses and manufacturers face product rejections, regulatory penalties, and costly recalls every year – not because their product is unsafe, but because their formulation was mostly never properly reviewed for compliance. Here in this blog you can get the exact solutions of your all concern.
Whether you manufacture packaged foods, dietary supplements, nutraceuticals, health supplements, or functional foods, an Expert Food Formulation Review and a thorough food ingredient assessment are non-negotiable steps in your regulatory journey. In this detailed blog, we will walk you through everything you need to know –
- What these services mean,
- Why they matter,
- What the process looks like, and
- How your food businesses manage this with confidence.
What Is Food Ingredient Assessment and Food Formulation Review?
To understand why these services are critical, let us first break down each term clearly:
Food Ingredient Assessment
Food ingredient assessment is a structured regulatory and scientific evaluation of each individual ingredient in your food or supplement product. It verifies whether every ingredient – including active components, additives, preservatives, colours, flavours, vitamins, minerals, enzymes, or novel ingredients is:
- Legally permitted under applicable regulations (e.g., FSSAI Food Safety and Standards Act 2006, Codex Alimentarius standards)
- Used within permissible limits and concentration levels
- Safe for the intended consumer population (adults, children, pregnant women, etc.)
- Sourced from approved and compliant raw material suppliers
- Not banned, restricted, or flagged as a novel/non-specified food ingredient without prior approval
Food Formulation Review
Food formulation review is a broader, holistic evaluation of your product’s entire formula. It assesses how ingredients interact with each other, validates nutritional composition against regulatory benchmarks, and ensures the full formulation aligns with the product category standards, labelling requirements, and health or nutrition claim permissions. It is, very important, a compliance audit of your recipe.
Nutraceutical Formulation Review and Assessment
For manufacturers producing health supplements, dietary supplements, nutraceuticals, and functional foods, a specialised nutraceutical formulation review and assessment is required. This goes a step further – examining dosage compliance, RDA (Recommended Dietary Allowance) adherence as per ICMR guidelines, and ensuring alignment with FSSAI’s Health Supplements, Nutraceuticals, and Special Purpose Foods Regulations.
“A compliant formula is not just a legal requirement – it is your product’s passport to the market, consumer trust, and long-term business sustainability.”
Why Food Businesses Cannot Afford to Skip Formulation Review
Many food business owners treat formulation compliance as an afterthought – something to address only when a problem arises. This is one of the most expensive mistakes in the food industry.
Here is why proactive food ingredient assessment and food formulation review is indispensable:
1. Legal Compliance
FSSAI’s FSS Act 2006 mandates that all ingredients and formulations meet prescribed standards. Non-compliance leads to product recalls, fines, and license cancellation.
2. Consumer Safety
Unassessed ingredients can cause adverse reactions. A thorough ingredient safety assessment protects consumers and shields your brand from liability.
3. Faster Market Entry
Pre-reviewed formulations sail through FSSAI registration and food facility licensing – cutting your time-to-market by weeks or even months.
4. Accurate Labelling
Your nutritional label, ingredient declarations, and health claims must align with your reviewed formulation – a prerequisite for FSSAI-compliant labelling.
5. Export Readiness
If you plan to export to the US, EU, or GCC markets, your formulation must meet international standards – FDA, EFSA, and Codex Alimentarius requirements.
6. Investor & Retail Confidence
Retailers, investors, and distributors require documented compliance proof. A reviewed formulation adds credibility and unlocks distribution opportunities.
The Regulatory Framework: What Governs Food Formulation in India?
India’s food regulatory landscape is primarily governed by the FSSAI, established under the FSS Act, 2006. Every food business owner and manufacturer must understand the key regulations that directly affect Food Formulation Review and Ingredient Assessment:
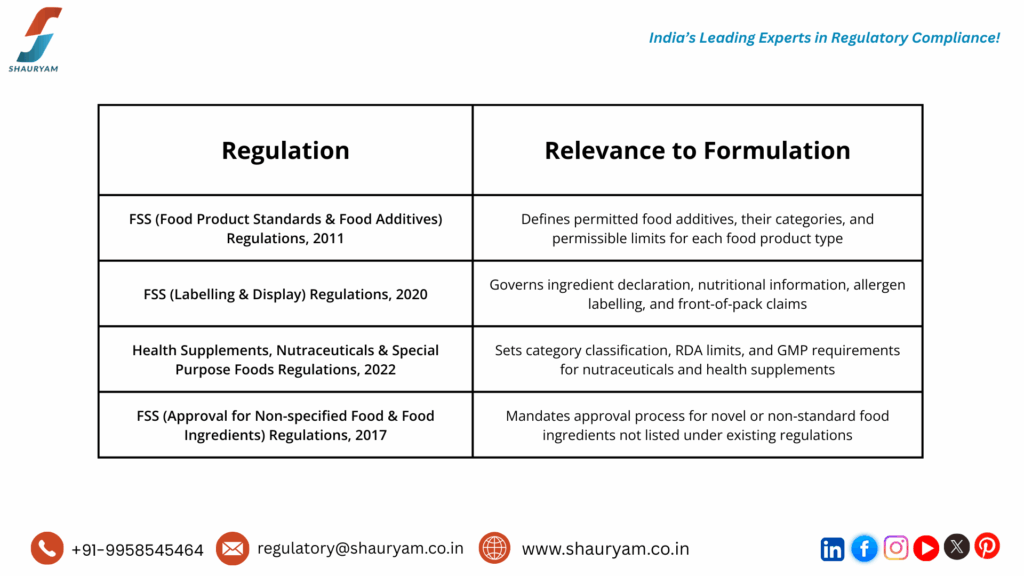

Understanding these FSSAI regulations and interpreting them correctly for your specific food or nutraceutical product category is complex. That is why partnering with an Expert for Food Ingredient Assessment and Food Formulation review is not just smart – it is very crucial for your business.
What Does Expert Food Formulation Review Cover?
A comprehensive expert food formulation review conducted by Shauryam covers every critical dimension of your product’s compliance. Here is what the review entails:
1. Ingredient Permissibility Check
We verify each ingredient against FSSAI’s positive lists of permitted food substances, additives, colours, flavours, vitamins, preservatives, enzymes, and minerals. If by any chance ingredients not found on the positive list require a separate novel food or non-specified food approval from FSSAI before the product can be marketed legally.
2. Dosage and Concentration Compliance
Even permitted ingredients have maximum and minimum usage limits. Our food ingredient assessment expert ensures that every additive, active ingredient, vitamin, and mineral in your formulation falls within the prescribed concentration limits – per serving, per 100g, or per 100 kcal as applicable to your category.
3. Product Category Classification
The regulatory requirements for a “dietary supplement” differ significantly from those for a “functional food,” a “fortified staple food,” or a “food for special dietary use (FSDU).” identifing the correct product category under FSSAI regulations, which then determines the entire compliance framework applicable to your product.
4. Nutritional Composition Assessment
We assess the nutritional profile of your formulation against FSSAI and ICMR benchmarks, including energy, protein, carbohydrates, fats, fibre, vitamins, and minerals. This is particularly critical for nutraceuticals, health supplements, infant foods, sports nutrition, and medical foods (FSMP).
5. Interaction and Compatibility Review
Ingredients do not always behave independently. Some nutrients interact with others, affecting bioavailability, stability, or safety. Our nutraceutical formulation review and assessment examines ingredient interactions to flag any formulation-level risks before production begins.
6. Claims Alignment Review
Health, nutrition, and functional claims on your packaging must align directly with your verified formulation. We review your intended claims against FSSAI’s permitted claims list and advise on substantiation requirements.
7. Label Declaration Readiness
The final output of our food formulation review includes a label-ready ingredient declaration list, nutritional information panel data, and guidance on mandatory declarations – all derived directly from your reviewed formulation.
Shauryam’s Formulation Review: Key Deliverables
- Ingredient permissibility report with regulatory references
- Dosage compliance assessment against FSSAI and ICMR limits
- Product category classification report
- Nutritional composition validation
- Ingredient interaction and safety flag report
- Claims assessment and substantiation guidance
- Label content recommendations for FSSAI-compliant packaging
- Action plan for non-compliant ingredients or concentrations
Our Step-by-Step Food Formulation Review Process
At Shauryam, we follow a structured, transparent process to deliver thorough food ingredient assessment and food formulation review for every client – from small food startups to large-scale manufacturers.
1- Initial Consultation and Product Brief
We begin by understanding your food or nutraceutical product concept, target market, intended consumer group, production scale, and business goals. This helps us scope the review accurately and identify the applicable regulatory framework from the outset.
2- Formulation Data Collection
You share your complete formula with us – including all ingredients, their quantities, sources, and supplier details. All information is handled under strict confidentiality.
3- Ingredient-by-Ingredient Assessment
Our regulatory experts conduct a detailed food ingredient assessment
– checking each ingredient’s legal status, permissible limits, source compliance, and interaction potential against FSSAI, ICMR, and Codex standards.
4- Formulation-Level Compliance Review
We assess the complete formulation for category compliance, nutritional benchmarking, and claims alignment. For nutraceutical and supplement products, we apply the nutraceutical formulation review and assessment framework, including RDA verification.
5- Gap Analysis and Compliance Report
We identify gaps, flag non-compliant ingredients or concentrations, and deliver a detailed compliance report with actionable recommendations. Where re-formulation is required, we guide you on permissible alternatives.
6- Re-formulation Support (If Required)
If your current formulation needs adjustment, our team actively supports re-formulation to achieve compliance without compromising your product’s intended functionality, taste, or effectiveness.
7- Final Compliance Sign-off
Once the formulation meets all requirements, we issue a compliance summary that supports your FSSAI registration, label development, and manufacturing documentation.
Who Needs Food Ingredient Assessment and Food Formulation Review?
If your business falls into any of the following categories, our expert food formulation review service is directly relevant to you:
- Food Manufacturers and Processors: Packaged food producers, FMCG companies, contract manufacturers, and private-label brands who need formulation sign-off before production scale-up
- Nutraceutical and Dietary Supplement Brands: Companies producing vitamins, minerals, herbal extracts, protein supplements, probiotics, or sports nutrition products
- Health Supplement Startups: Early-stage brands building their first compliant product before approaching investors or retail distributors
- Food Importers: Businesses importing food or supplement products into India who need to validate foreign formulations against Indian FSSAI standards
- Functional Food Brands: Companies adding health-functional ingredients (e.g., fortified beverages, enriched cereals, probiotic dairy) to mainstream food products
- Organic and Natural Food Brands: Businesses using plant-based, herbal, or botanical ingredients that require specific compliance verification
- Food Exporters: Indian manufacturers seeking to export products to the US, EU, GCC, or Southeast Asian markets, requiring multi-jurisdictional formulation compliance
- Contract Development and Manufacturing Organisations (CDMOs): Facilities that manufacture for third-party brands and need formulation-level regulatory sign-off
Common Formulation Mistakes That Lead to FSSAI Rejection
Through years of supporting food businesses, our compliance regulatory expert team at Shauryam has identified the most frequent formulation errors that lead to product rejection, regulatory action, or market withdrawal.
Awareness of these mistakes is your first line of defence:
- Using non-permitted additives or colours: especially synthetic colours not on FSSAI’s approved list, or additives used beyond their permitted categories
- Exceeding vitamin or mineral upper limits: a common issue in nutraceutical and health supplement formulations where high-potency levels are used without verifying ICMR or FSSAI upper limits
- Incorrect product classification: labelling a product as a “dietary supplement” when the ingredients and dose format technically qualify it as a drug under the Drugs and Cosmetics Act
- Using novel or non-specified ingredients without prior approval: ingredients not listed under FSSAI’s existing standards require separate approval, which many brands skip
- Formulating for a foreign market standard: using US FDA or EU standards as the compliance benchmark without checking FSSAI-specific requirements
- Claims not supported by the formulation: making health or nutrition claims on the label that the actual formulation cannot substantiate
- Allergen declaration errors: failing to properly declare major allergens (gluten, milk, soy, nuts) in the ingredient list as required under FSSAI Labelling Regulations 2020
“A formulation review done at the concept stage costs a fraction of a product recall done at the distribution stage.”
Special Focus: Nutraceutical Formulation Review and Assessment in India
The nutraceutical and health supplement segment in India is witnessing explosive growth, driven by rising health consciousness and post-pandemic awareness. However, it is also one of the most heavily scrutinised product categories by FSSAI, particularly following the 2022 Nutraceuticals Regulations and ongoing inter-ministerial discussions about the regulatory boundary between food and drugs.
A specialised nutraceutical formulation review and assessment is essential for:
- Verifying correct classification of your product under FSSAI’s six functional food categories as per the 2022 Nutraceuticals Regulations
- Ensuring vitamin, mineral, and amino acid dosages comply with ICMR RDA limits and FSSAI’s upper permissible levels
- Checking that excipients, fillers, and dosage formats (tablets, capsules, powders, gummies) are permitted for food-category products
- Assessing whether disease risk reduction claims require drug-category classification, which the Inter-Ministerial Committee has been evaluating
- Verifying ingredient origin, extraction methods, and supplier compliance for botanical and herbal ingredients
- Confirming GMP (Good Manufacturing Practice) readiness under Schedule IV of FSSAI regulations
At Shauryam, our expert food formulation review for nutraceuticals is conducted by regulatory experts with deep knowledge of both food and nutraceutical compliance frameworks – ensuring your product lands in the correct regulatory lane and stays there.
Why Choose Shauryam, for Food Ingredient Assessment and Food Formulation Review?
At Shauryam, we combine updated regulatory expertise with a practical, business-first approach. We do not just identify compliance gaps – we help you build a compliant, market-ready product from the ground up.
Here is what sets our food formulation review service apart:


Conclusion
In India’s fast-evolving and increasingly regulated food market, getting your formulation right is not optional – it is the foundation of everything that follows. Food ingredient assessment and food formulation review are not bureaucratic formalities. They are the scientific and regulatory backbone that determines whether your product reaches the market safely, legally, and with the trust of consumers and regulators alike.
Whether you are a seasoned food manufacturer scaling a new product line or an entrepreneur launching your first food or nutraceutical brand, an expert food formulation review by Shauryam gives you the regulatory clarity, compliance confidence, and market readiness to move forward – without expensive shocks.
The Indian food market is full of opportunity. Make sure your formulation does not stand in the way of it.
Ready to Get Your Food Formulation Reviewed?
Let Shauryam’s experts handle your food ingredient assessment and food formulation review – so your product is compliant, market-ready, and built to last.
Talk to our regulatory expert today.
Food ingredient assessment is a systematic regulatory and scientific evaluation of every ingredient in your food or supplement product. For food manufacturers in India, this means checking each ingredient – whether it is a food additive, colour, flavour, vitamin, mineral, botanical extract, or novel substance – against FSSAI’s approved lists and permissible limit regulations. Without this assessment, you risk using ingredients that are either banned in India, restricted to specific product categories, or used in quantities that exceed legal limits. Non-compliance can lead to product rejection, market recall, or FSSAI enforcement action – all of which are far more expensive than a proactive ingredient assessment.
Food testing and food formulation review are two distinct but complementary activities. Food testing is a laboratory analysis that confirms what is actually present in your product at a given concentration – it measures and detects. Food formulation review, on the other hand, is a pre-production regulatory assessment of your intended recipe. It confirms whether your formula is legally compliant, appropriately categorised, and aligned with label declarations before you manufacture a single unit. Think of it this way: testing tells you what you have; formulation review tells you whether what you plan to make is legally permissible. Both are necessary, but a formulation review should ideally come first.
Ideally, food ingredient assessment and food formulation review should be completed before FSSAI registration – and certainly before production begins. Here is why: the FSSAI licensing and product registration process requires that your product’s formulation, ingredients, and labelling already meet all applicable standards. If non-compliance is discovered during or after registration, you will face delays, additional costs, re-formulation, and potential business disruption. Getting a formulation review at the product development stage is the most cost-effective approach. For businesses that have already launched, a formulation review is still valuable as a compliance audit to identify and resolve any risks before FSSAI inspections or retail audits occur.
There is significant overlap, but nutraceutical formulation review and assessment has additional layers of complexity. For nutraceuticals, dietary supplements, and health supplements, the review must go beyond basic ingredient permissibility to include: verification of dosage against ICMR Recommended Dietary Allowances (RDAs), assessment of whether the product falls under food or drug regulatory jurisdiction (a critical distinction under India’s Inter-Ministerial Committee review framework), checking for GMP (Schedule IV) compliance requirements, and evaluation of claims for permitted health or nutrition status. The FSSAI’s Nutraceuticals Regulations, 2022 classify health supplements into six functional food categories, each with distinct compliance requirements. A generic food formulation review will not address these nuances – you need a specialist review from experts who understand both the food and pharmaceutical regulatory frameworks in India.
If a food ingredient assessment identifies an ingredient as a novel or non-specified food substance under FSSAI regulations – meaning it does not appear on FSSAI’s positive lists and does not have established standards – you cannot legally use it in your product without obtaining prior approval from FSSAI under the FSS (Approval for Non-Specified Food and Food Ingredients) Regulations, 2017. This approval process requires submitting a comprehensive dossier with safety data, intended use, and scientific justification. At Shauryam, when our formulation review flags a novel ingredient, we advise you on the best course of action – which could include either pursuing the approval dossier process or substituting the ingredient with a compliant alternative that achieves similar functional outcomes in your product.
Yes, absolutely. An expert food formulation review can be scoped to cover both Indian FSSAI requirements and the regulatory requirements of your target export markets simultaneously. For example, if you are planning to export to the United States, your formulation must comply with FDA regulations under 21 CFR, including the GRAS (Generally Recognised as Safe) standard for additives and dietary ingredient notification requirements. For EU exports, formulations must align with EC Regulation 1333/2008 on food additives and the European Food Safety Authority (EFSA) guidelines. At Shauryam, we offer multi-jurisdiction formulation review services, allowing you to build a product that is compliant across multiple markets from day one – saving significant time and cost compared to conducting separate country-by-country reviews later.
The timeline for a food ingredient assessment and food formulation review depends on the complexity of your product – specifically the number of ingredients, the product category, whether any novel ingredients are present, and the number of markets you need compliance for. For a straightforward packaged food product with standard ingredients, a formulation review can typically be completed within 5 to 10 working days. For nutraceutical, health supplement, or functional food products with multiple active ingredients, complex interactions, or multi-jurisdiction scope, the review may take 2 to 4 weeks. At Shauryam, we always discuss timelines during the initial consultation and agree on a delivery schedule upfront – so your product development planning is not disrupted.
A food formulation review should be conducted every time there is a meaningful change to your product’s formulation. This includes adding a new ingredient, changing the concentration of an existing ingredient, substituting a supplier (which may change ingredient specifications), or modifying the product category. Additionally, even if your formulation does not change, periodic compliance re-reviews are advisable when regulations are updated – which happens regularly with FSSAI. For example, FSSAI’s nutraceutical regulations were significantly revised in 2022, meaning products that were compliant before may have needed re-evaluation. At Shauryam, we recommend an annual compliance review as a best practice, particularly for nutraceutical and health supplement manufacturers, to stay current with regulatory changes and avoid any lapses in compliance.





